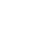Science in Support of National Objectives
Combustion of hydrocarbon fuels provides the majority of the energy used in the United States for power generation, transportation, and industrial processes. Dependence on imported hydrocarbon fuels is a major component of the US international balance of payments, with significant impacts on the financial and energy security of the United States. Emissions from these power systems make dominant contributions to global climate change and other environmental challenges world-wide. LLNL combustion chemistry research is directed towards understanding the details of these processes, in both qualitative and quantitative terms, leading to improved efficiency and reduction in emissions to the atmosphere. Current work is directed towards understanding and predicting the possible impacts of alternative fuels, including fuels derived from biological systems and new geological sources, such as oil shale and oil sands. The chemical kinetic reaction mechanisms developed at LLNL are used widely in the United States and many foreign groups, and dissemination of these mechanisms via this web page is a major priority of the LLNL Combustion Chemistry Group.
Major Accomplishments
Combustion chemistry has been pursued at LLNL since about 1975, and many significant advances can be attributed to this work. Early studies addressed major sources of unburned hydrocarbon emissions from internal combustion engines by a variety of flame quenching mechanisms. We established the role of volume bulk flame quenching in stratified charge engines. We also showed that flame quenching on cooled walls of engine combustion chambers was not the major source of hydrocarbon emissions from spark-ignition (SI) engines, overturning 50 years of engine design lore.
Later, we were able to demonstrate the major role of molecular structure of different hydrocarbon fuel molecules on their rates of ignition in SI and diesel engines, which in turn determines the octane and cetane ratings of fuels in these engines. These fuel rating systems had been determined empirically from engine experiments in the 1920s and 1930s, but no scientific basis for the ratings had been established. Our work at LLNL focused on the low temperature reaction pathways that oxidize these fuels, showing the specific chemical reactions that lead to the differences between knocking tendencies of fuels with different sizes and molecular structure. This work was recognized scientifically and awarded the 1991 Horning Memorial Award by the Society of Automotive Engineers and the 1993 Thomas Midgley Award from the American Chemical Society.
More recently, we have given considerable attention to hydrocarbon combustion in diesel and HCCI engine systems. We have shown how diesel ignition varies with fuel molecular size and structure, and how these factors determine the subsequent production of soot and its undesirable emissions from diesel engines. This work was recognized by the 2000 Arch Colwell Award of Merit from the Society of Automotive Engineers, providing an overall understanding of the role of fuel structure on ignition and soot production in diesel engines. Our later kinetic modeling explained observations from diesel engine studies in which small amounts of additives containing oxygen atoms (e.g., alcohol, ketone, or ether species) sharply reduced soot emissions. This work received the 2003 Arch Colwell Award of Merit from the Society of Automotive Engineers.
Additional accomplishments by our group have established the role of detailed chemical kinetics in the behavior of a wide variety of other combustion systems. We provided a kinetic framework for determining the role of molecular size and structure on detonation properties via the prediction of detonation cell sizes, on stability of pulse combustion systems, on combustion in supercritical water, and in selected explosives. We also have made significant contributions to the theory of inhibition of flames and ignition via kinetic interactions with halons such as HBr and CF3Br, as well as organophosphorus compounds such as dimethyl methyl phosphonate (DMMP) and others, and we have developed the first kinetic models for selected toxic organophospates as well.
Recently, our work has been focused on development of surrogate fuel models to represent gasoline fuels, diesel fuels, and their mixtures with alternative fuels (e.g., biofuels). These fuel models and their mechanism reductions enable the simulation of fuel effects in engine simulations of conventional and advanced engine combustion.